In the United States there are many regulatory requirements at both the state and federal levels regarding the discharge of wastewater from industrial manufacturing facilities. Any company that discharges any sort of effluent into sewer systems, lakes, streams or the ocean is required to neutralize this effluent before discharge.
Noncompliance can result in financial penalties, at a minimum, and may also include other consequences such as being required to remediate any resulting environmental damage. It is in the best interests of any company—and depending upon local rules, it may be compulsory—to record discharged effluent characteristics such as pH at the point where it leaves their facility.
pH Monitoring and Neutralization Systems
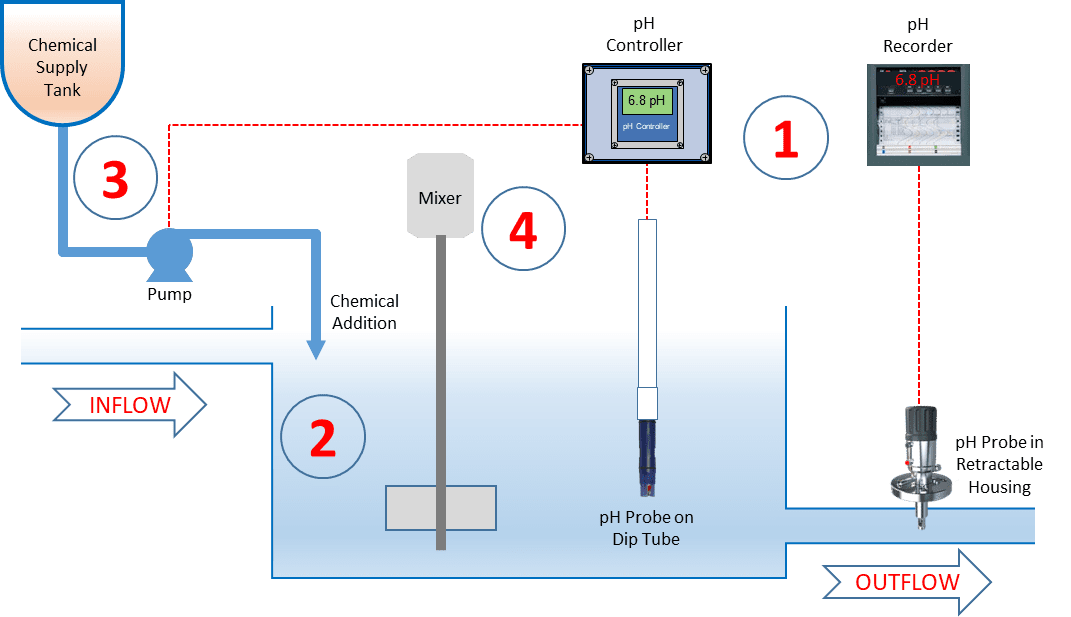
Many plants operate an automatic pH neutralization system that monitors and controls effluent pH by chemical addition. A simple pH neutralization system consists of four basic components:
- Instrumentation for monitoring, controlling, and recording, including pH and ORP electrodes
- Effluent holding tank
- Chemical reagent storage tanks and addition pumps
- Agitator(s)
In such a system, effluent flows into the holding tank where a pH sensor (also known as a pH electrode or pH probe) measures the pH of the solution. The pH controller has a preset pH set point for the effluent and uses this measured value to determine whether the effluent is within compliance with that set point. If not, chemical pump(s) are operated to inject acidic or caustic solutions as required to bring the effluent in the holding tank to the correct level. The agitator(s) keep the contents of the holding tank adequately mixed, so that the pH probe is always measuring a representative sample of the effluent and any chemicals added are properly and quickly distributed within the tank.
The pH Neutralization Process
pH neutralization can be accomplished in a batch or continuous mode. Batch systems operate by filling an effluent holding tank and carefully treating the contents with a variety of chemicals to remove metals and other contaminants before finally setting the pH level before discharge. Continuous systems operate by flowing effluent through a series of tanks or basins where measurements are taken and treatment chemicals added. Whichever treatment mode is used, it is of primary importance that measurements can be relied upon and chemical addition equipment is in good condition if the final effluent is to be safe to discharge.
After the neutralization process is completed, the effluent is discharged to waste. At the point of discharge, a pH sensor should be installed dedicated to monitoring and recording the effluent’s pH level. This provides a record of the discharge pH level that can be produced for inspection by external agencies while also providing valuable data for internal use.
In many industrial plants, maintenance of wastewater treatment equipment can be a key challenge. In these plants, wastewater is often a cost center rather than a revenue generating center within the business, and therefore does not enjoy the maintenance resources of the rest of the facility. Poorly operating pH measurement systems can lead to off spec neutralization. In these cases, manual override of dosing systems is a common practice, and costly overdosing with correction chemicals can occur.
Common Problems for pH Neutralization Systems
Many of the operational problems encountered in a pH neutralization system are related to pH sensor performance, with incorrect choice of electrode technology and placement being common causes. pH sensor selection is therefore a key element in problem-free operation.
Sensor Selection
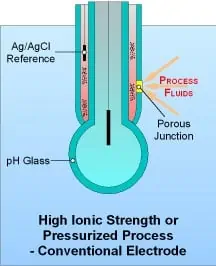
Single junction combination sensors combine a pH glass electrode with an Ag/AgCl electrode-in-KCl reference cell in a single convenient package, and are available in a variety of sizes. The reference junction in these electrodes is porous and allows the KCl salt solution to come into direct contact with the effluent, thus completing the sensor electrical circuit. These electrodes are depleting, in that the internal KCl reservoir will become diluted over time and is susceptible to poisoning when the measured effluent contains heavy metals, sulfides, proteins, or other materials that interact with silver. Dilution causes measurement drift, while poisoning causes measurement error and eventual electrode “death.” Furthermore, should the porous junction become blocked, proper measurement is not possible. A sign of junction blockage is sluggish sensor response, which can lead to expensive overdosing of pH correction chemicals. Without proper maintenance, pH measurement systems using 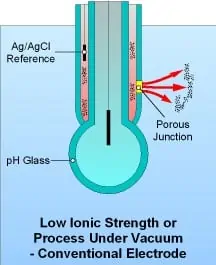
Double junction combination sensors are similar to single junction electrodes, except that they have two porous junctions in series instead of one. They typically have a longer lifetime than single junction sensors because they generally have better resistance to poisoning. They still require frequent maintenance and standardization if the pH values they produce are to be reliable.
Differential pH electrodes were designed to combat the poisoning of the Ag/AgCl electrode in standard reference cells. In a differential electrode, two pH glass electrodes are used, one in contact with the effluent and the other immersed in KCl within the probe body (the reference junction). While this combats the problem of poisoning, there 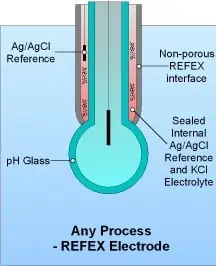
Non-porous reference junction pH electrodes (such as the REFEX brand) have a glass pH sensor just like all others. The difference is that they do not have a porous reference junction, so poisoning and dilution of the reference junction electrolyte is prevented. The lifespan of non-porous junction electrodes in wastewater neutralization systems is much, much longer than those with porous junctions, and maintenance requirements are almost zero. Typically, non-porous junction electrodes require only periodic cleaning as a maintenance requirement, and this can be easily automated for “hands-free” operation.
Mounting Considerations
pH sensors used in wastewater neutralization systems should be suitable for the use. As many systems are open tank, pH sensors are often installed as dip tubes, with a plastic pipe connected to the back of the pH sensor over the sensor wire. A holding bracket of some kind is used to secure the dip tube in place. For closed top effluent tanks, sensors can be installed through entry ports either in the top of the tank or through the side. When mounting, it is recommended that the following be considered:
- The sensor dip tube must be properly supported to prevent the sensor from moving around in the tank. Excessive movement may cause impact of the sensor on the tank wall or agitator, leading to damage.
- Ensure the electrode is installed with a moving, turbulent flow around it. This will help minimize coating and therefore reduce maintenance overhead.
- Make sure the electrode is not mounted where chemical addition takes place, as this will unduly influence or bias the measurement. It can take a while for mixing and reaction to occur in a tank.
- In applications with heavy coating, consider an automated sensor cleaning system. For dip tube installations, a spray head attachment fed with a cleaning agent suitable for the process—often clean water, but other materials such as detergent are sometimes used—is a good option. For closed tank mounted probes, an automatic retractor and cleaning system like the examples from Exner Process Equipment below can be used.
Electrode Maintenance
Common problems experienced with pH electrodes (and recommended solutions) are:
- Buildup of oils and/or solids on pH electrodes, necessitating frequent cleaning: The lifespan of electrodes in these applications tends to be quite short. Use sensors with non-porous reference junctions, as these have much greater resilience to this kind of problem than porous junction electrodes. The addition of an automated cleaning system will reduce maintenance requirements further.
- pH bulb breakage or failure as a result of abrasives or heavy solids in the effluent: Fit probes with a bulb guard or house in a stilling well to slow the flow of material over the pH electrode and protect it from abrasion or impact from large objects.
- Reference junction fouling or plugging: Use non-porous reference junction electrodes to prevent reference junction blockage.
- Short electrode life caused by reference poisoning due to interaction between silver ions in the electrode and material in the effluent: Use non-porous reference junction electrodes to completely remove the possibility of reference junction poisoning.
Electrode Care and Cleaning
Whenever handling pH electrodes and any associated solutions, always wear gloves and safety glasses. Safety first.
Keep all electrodes wetted when not in use. Electrodes will become useless if allowed to dry out. Electrodes will typically be supplied fitted with a “boot” or bottle filled with a salt solution from the manufacturer and this should not be removed until the electrode is placed into use. A good tip is to keep the boot or bottle to one side so it is available should the electrode be taken out of service. Fill with a pH 7.0 buffer when putting back on the probe if storage solution is not to hand.
A common way to clean electrodes is to immerse it in a mild (2% to 5%) solution of hydrochloric acid (HCl). This will dissolve most coatings. Always make sure the solution used for cleaning is compatible with the process—sometimes other solutions are more effective. Soak the sensor for 5 minutes then rinse with water. For heavy coatings, it may be necessary to repeat several times, carefully wiping with a soft, non-abrasive cloth between immersions. Never use abrasive materials or brushes to clean pH sensors, as this can damage the measuring surface and render the electrode useless.
Conclusion
pH neutralization is a critical wastewater treatment process in many industrial manufacturing environments. Through proper choice of sensor components, ongoing operation of the system can be optimized resulting in minimal maintenance and significantly lowering costs and risk of accidental discharge.
Images used by kind permission from: